A Fully Integrated, Patient-Centric Approach
Mavens combines strategy, technology, and partnerships to provide outcome-driven, patient-centric solutions for Life Sciences.
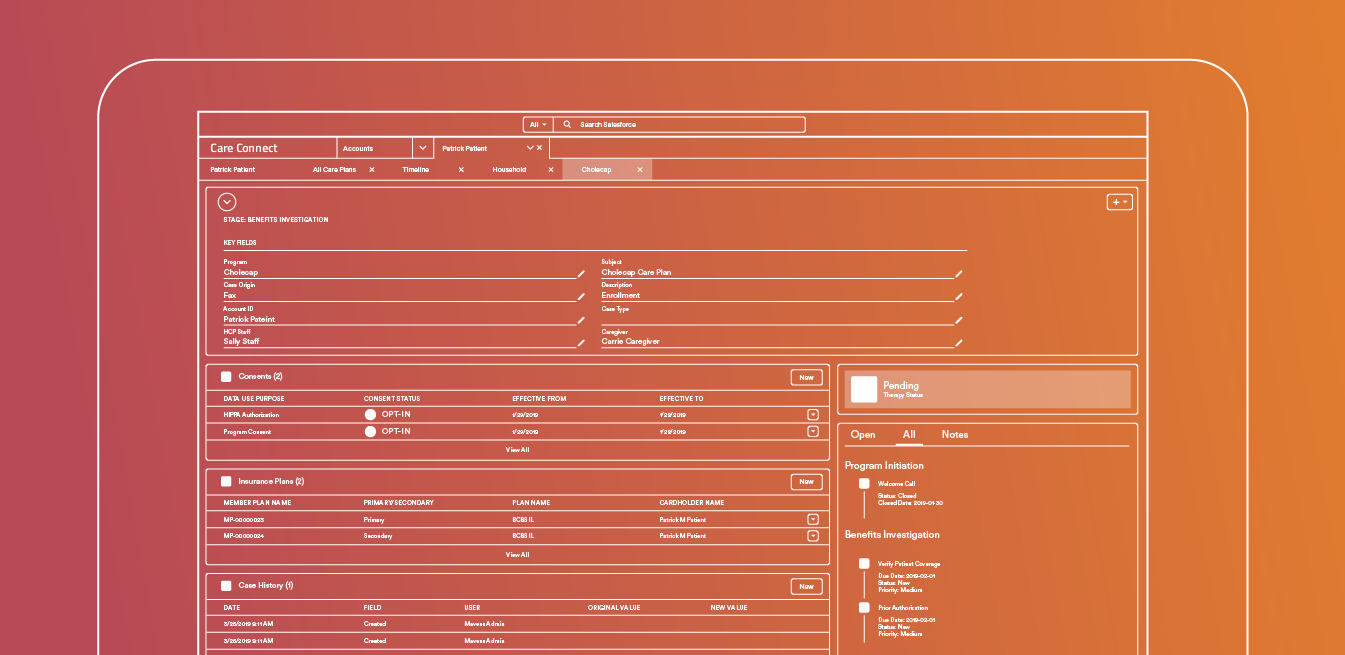
Care Connect
Orchestrate complex patient journeys across teams and channels with one flexible, secure platform.

Solving Life Sciences toughest challenges.
Our products are powered by Salesforce to provide security and flexibility for the future of Life Sciences.